|
Ruxolitinib was tapered further to 10 mg once daily for 3 months, and the patient currently receives 10 mg every other day without loss of efficacy, treatment adverse effects, or laboratory-result abnormalities. The dosage was subsequently tapered to 10 mg twice daily for an additional 8 months ( Fig 1 C and D) without loss of efficacy. After 4 months, he had a nearly complete hair regrowth of eyebrows and scalp ( Fig 1, B). According to the body surface area Dubois formula for 25 mg/m 2 per dose in a patient with a height of 1.4 m and weight of 80 kg, the calculated dose was 40 mg ruxolitinib, and the patient began receiving ruxolitinib 20 mg twice daily. After consent and baseline laboratory studies (complete blood cell count, liver function tests, serum electrolytes, renal function, and lipid panel), we opted to start at a dose of 25 mg/m 2. 9 A discussion of potential treatment-related adverse effects with the patient and his mother ensued and included those listed earlier, as well as hypertriglyceridemia and vascular occlusion or cardiovascular stroke. Toxicities were similar across doses and included elevation of liver enzyme levels, creatinine-level elevation in the setting of acute kidney injury, anemia, lymphopenia, leukopenia, neutropenia, and thrombocytopenia. 9 In this study, oral ruxolitinib was administered twice daily in escalating doses levels of 15, 21, 29, 39, and 50 mg/m 2 per dose. For dosing, we followed a phase 1 study with dose escalation of ruxolitinib in patients with hematologic malignancies from aged 2 to 22 years. Screening laboratory test results for HIV, hepatitis B and C, and tuberculosis were negative before the initiation of treatment. After denial of tofacitinib by insurance, ruxolitinib was approved and initiated with careful monitoring for hematologic effects. Given lack of response to previous treatments and severe psychological distress, we initiated JAKI therapy. By 8 months after his initial presentation, his disease had progressed to involve his entire scalp and eyebrows, consistent with alopecia totalis ( Fig 1, A). 
Within 2 months, he had developed several additional patches on his scalp and was treated again with intralesional triamcinolone, clobetasol solution, and oral prednisone, without response. 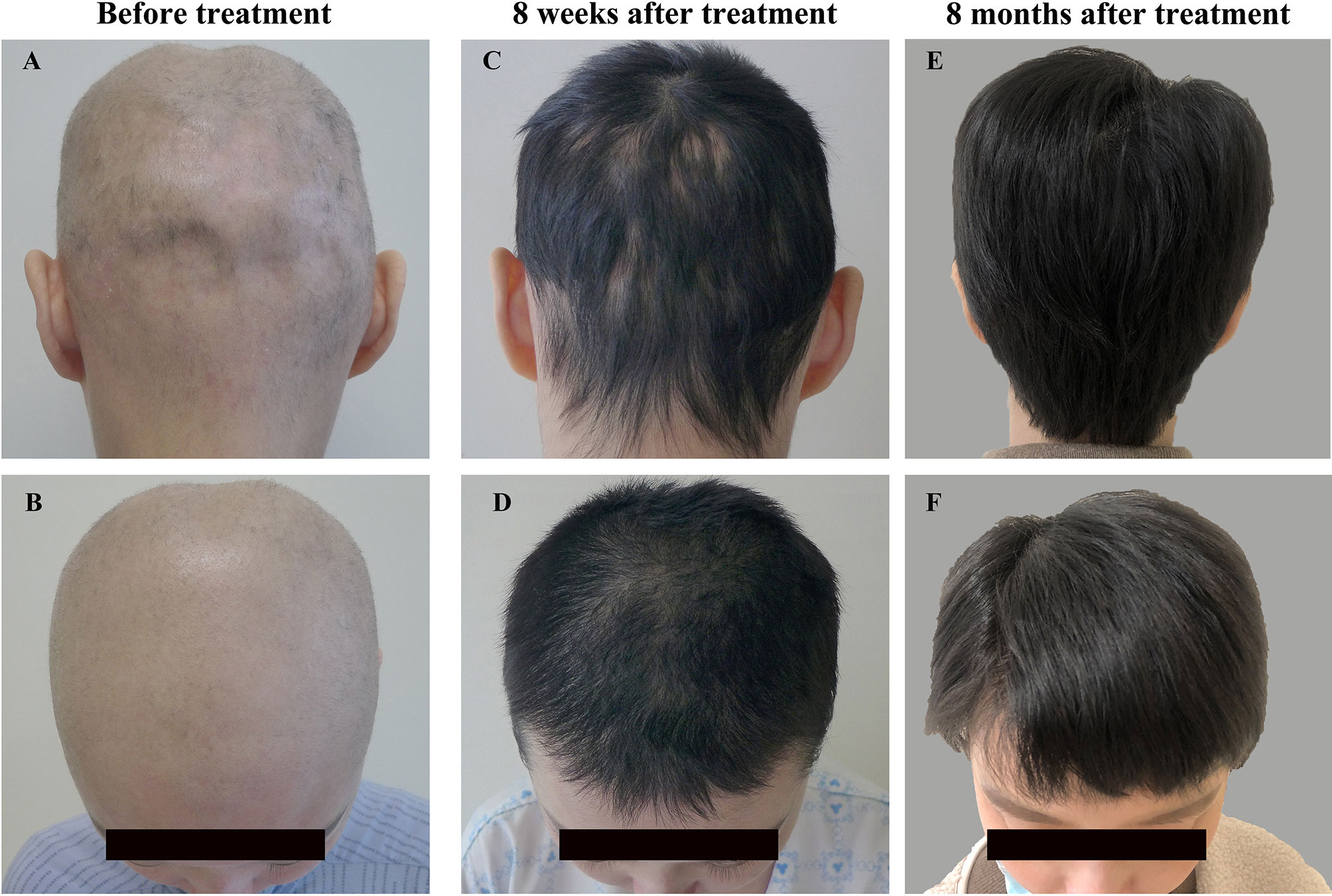
It is the most severe form of alopecia areata, and as such is quite rare, occurring only in around 1 in 100,000 people.Īs well as the three types of alopecia outlined above, there are a couple of other ways alopecia can be classified.A 9-year-old obese boy first presented with a single patch of alopecia areata on his scalp that did not respond to intralesional triamcinolone. This term relates to hair loss all over the body, meaning that the entire epidermis is free from hair. It is a more advanced stage of alopecia areata, which progresses to totalis and universalis in around 1 – 2 per cent of cases. This is hair loss affecting the whole of the head, including eyelashes and eyebrows. This is one of the most commonly reported types of hair loss, referring to bald patches or spots on the scalp. 
But what is the difference between the three? However, what they don’t always know is that there are three main types of the condition – alopecia areata, alopecia totalis and alopecia universalis. Most people know alopecia to be a form of hair loss.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
